instaCELL Cytotoxicity Assay Kit
The instaCELL Cytotox assay kit is designed for labs that need flexibility without compromising reliability.
Simply keep your instaCELLs frozen and run your assay within 3 days. No prior cultivation required.
How the assay works:
Extracts from your medical device components are incubated with L-929 fibroblast cells for 24 hours. Cell viability is then measured using detection reagents, which are metabolized by living cells. Results are evaluated against a cytotoxic control, indicating your component's cytotoxicity.
Why instaCELL?
• L-929 assay ready instaCELLs perform comparably to cells from continuous culture
• Qualified kits deliver reliable, repeatable results with maximum scheduling flexibility
• Validated for biocompatibility testing of medical devices per ISO 10993-5
• Medical device reference materials are available separately
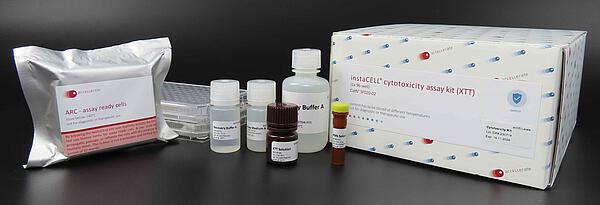
Each kit includes:
• Frozen L-929 instaCELLs
• Media and buffers
• Cytotoxic control
• Colorimetric (XTT) or fluorometric (resazurin) detection reagent
• Assay plates
special offer
Get assay ready – we are now offering a special validation package discount. Get 40% off of up to 10 kits that you order within 6 months. Perfect for running all your validations and getting your lab set up with reliable, hassle-free testing in no time!