The instaCELL TNF-α neutralization assay kit is used to determine the potency of tumor necrosis factor-alpha (TNF‑α) neutralizing/inhibiting therapeutics. These recombinant antibodies and soluble proteins are used to regulate excessive inflammatory responses in the body. A dysregulation of TNF-a is associated with diseases like Alzheimer's disease, cancer, major depression, psoriasis and inflammatory bowel disease (IBD).
The assay utilizes the cytotoxic effect of TNF-α on the well-established mouse fibroblast cell line L-929 to determine the inhibitory efficacy. Cells are incubated with a dilution series of a drug/TNF-α mixture, with a constant TNF-α concentration, and then analyzed via a resazurin cell viability assay. Based on a reference standard, which has a fixed, known effective unit size, the inhibitory antibody can be potentiated.
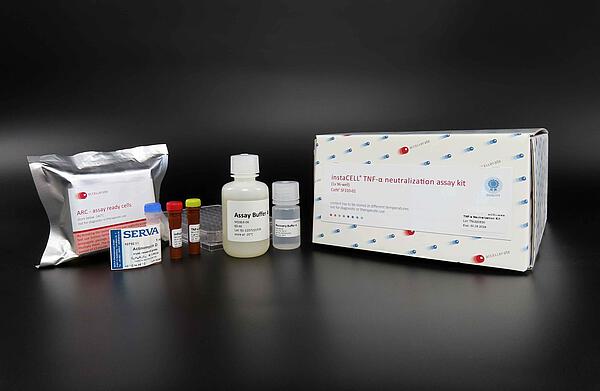
The instaCELL TNF-α neutralization assay kit includes prequalified assay-ready L-929 cells as well as media, reagents, reference standard, and 96-well plate to perform the assay according to the European Pharmacopeia. Assay Ready Cells are frozen cell aliquots which can be used directly in the assay without prior cultivation, basically like a reagent.